Drug Pricing Indexes: Understanding Maximum Fair Price (MFP)
Welcome to The Coy Pharmer, where our mission is to cultivate clarity in the often bewildering world of pharmacy benefits. Today we are unpacking Maximum Fair Price (MFP), one of the most consequential shifts in Medicare drug pricing in decades. Created under the Inflation Reduction Act, MFP introduces direct federal price negotiation for certain high-cost medications covered by Medicare. If you work anywhere along the drug supply chain… manufacturer, PBM, plan sponsor, pharmacy, broker, or policy… this policy will affect you. Even if you do not work in healthcare, MFP will influence premiums, taxes, innovation, and access over time. Let’s break down what MFP actually is, how it works, and why it matters.
DRUG PRICING INDEXES
2/19/20265 min read

In this Episode:
The Basics
Breaking down
the AcronymsProgram Inclusion
In Practice
Negotiation Timeline
The Fine Print
Pros and Cons for
ManufacturersPharmacy Impacts
Plan Sponsor Impacts
340B Program Interaction
Manufacturer Strategy
The Basics of MFP
MFP in Practice
Conclusion
Maximum Fair Price represents a fundamental shift toward government-directed pricing within Medicare. It introduces new rules, new incentives, and new uncertainties across the pharmaceutical ecosystem.
Whether MFP ultimately reduces costs without dampening innovation remains an open question. What is certain is that stakeholders who understand the mechanics will be far better positioned than those who do not.
At The Coy Pharmer, our goal is to turn complexity into clarity so decision-makers can act with confidence rather than confusion.
If you work anywhere along the drug supply chain and are trying to understand how these changes affect your strategy, benefits, or bottom line, you are exactly who this content is for.
Stay informed. Stay curious. And remember… knowledge is leverage.
Timeline and Negotiation Process
Maximum Fair Price is not a suggested price. It is a mandatory price for eligible Medicare utilization.
The result is a relatively small number of products that represent a disproportionately large share of Medicare drug spending.
This long runway creates both stability and rigidity. Stakeholders know what is coming, but they cannot easily adjust once decisions are locked in.
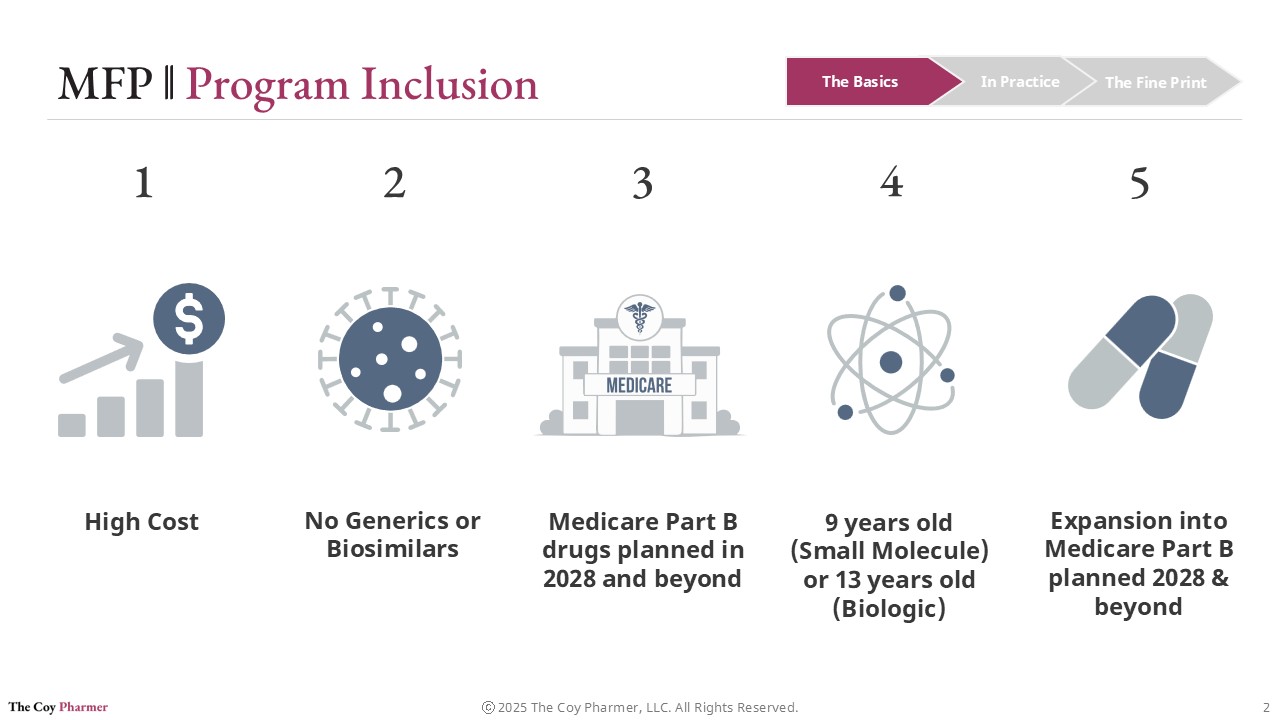
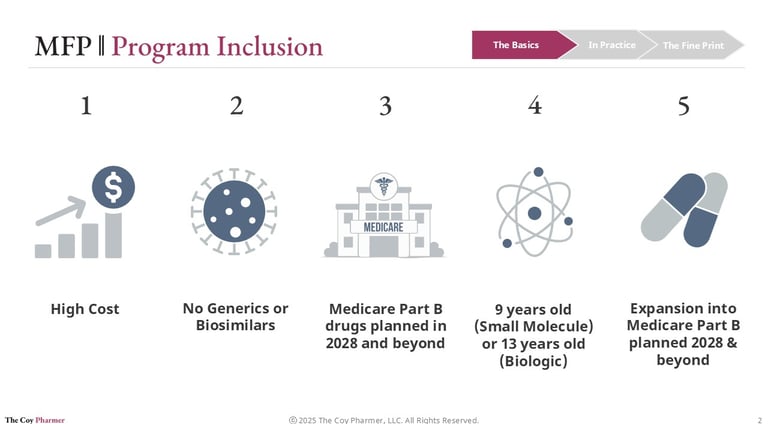
Maximum Fair Price is the government-negotiated price that Medicare will pay for select high-spend, brand drugs that lack generic or biosimilar competition.
Historically, Medicare Part D relied on private negotiation through PBMs. The federal government did not directly negotiate drug prices.
Not every drug qualifies for MFP. CMS selects medications based on statutory criteria designed to target products with the largest fiscal impact and limited market competition.
To be eligible, a drug generally must:
Be a high-cost drug in Medicare
Lack generic or biosimilar alternatives
Have been on the market 9-13 years, depending on molecule type
Meet specific regulatory definitions under the program
Initially, the program applies to Medicare Part D drugs. Medicare Part B drugs are scheduled to enter the negotiation process later, with implementation beginning in 2028.
Maximum Fair Price fundamentally changes that structure. The Centers for Medicare & Medicaid Services (CMS) now negotiates directly with manufacturers for specific drugs and establishes a ceiling price that must be offered to Medicare.
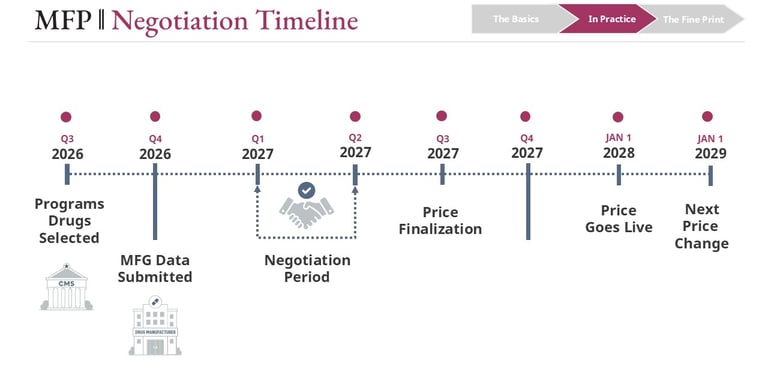
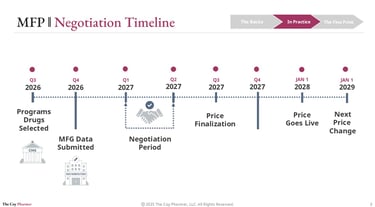
MFP implementation is not quick. It operates on a multi-year cycle that requires forecasting, modeling, and strategic planning across the entire industry.
A typical sequence looks like this:
Drug selection by CMS approximately two years before implementation
Manufacturer data submission including R&D costs, production expenses, and sales data
Negotiation period between CMS and the manufacturer
Final price announcement well in advance of activation
Implementation in Medicare plans
The Fine Print
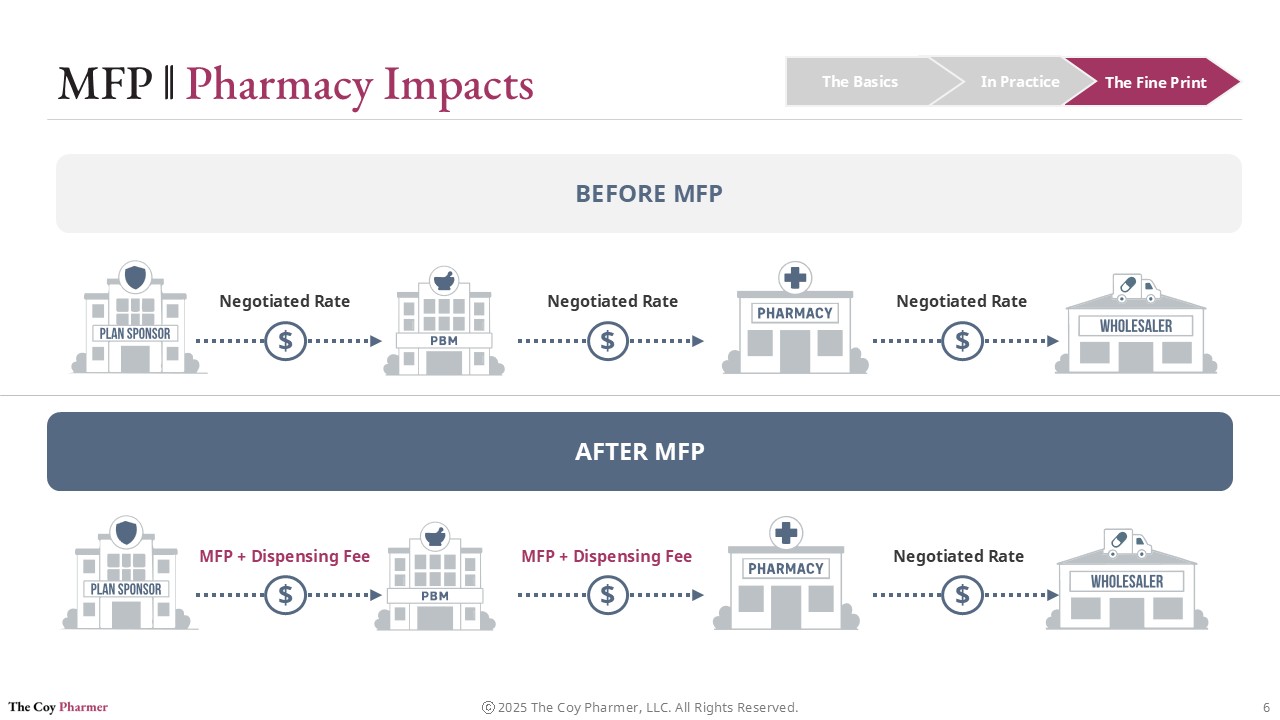
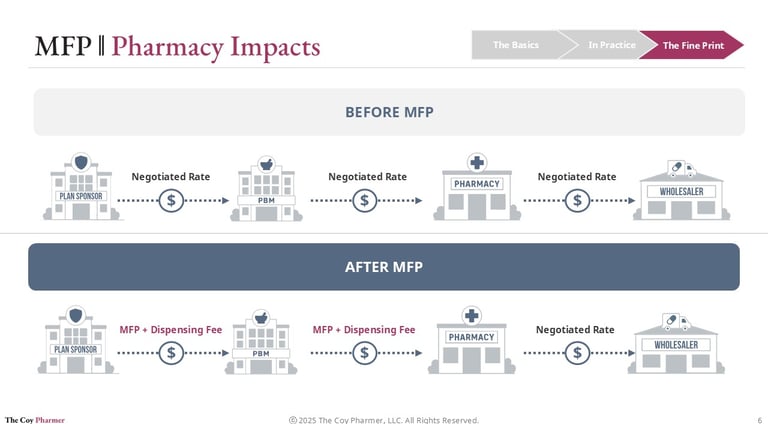
Impact on Pharmacies
Pharmacies operate at the downstream end of pricing decisions but often feel the effects first.
Under MFP:
Reimbursement structures may change
Dispensing margins could narrow
Contract terms with PBMs may shift
Inventory risk may increase for affected products
Pharmacies do not negotiate MFP directly. They must adapt to whatever reimbursement framework flows through plan sponsors and PBMs after the negotiated price is established. The MFP program does not necessarily improve the pharmacy’s ability to buy drugs, but may lower reimbursement rates.
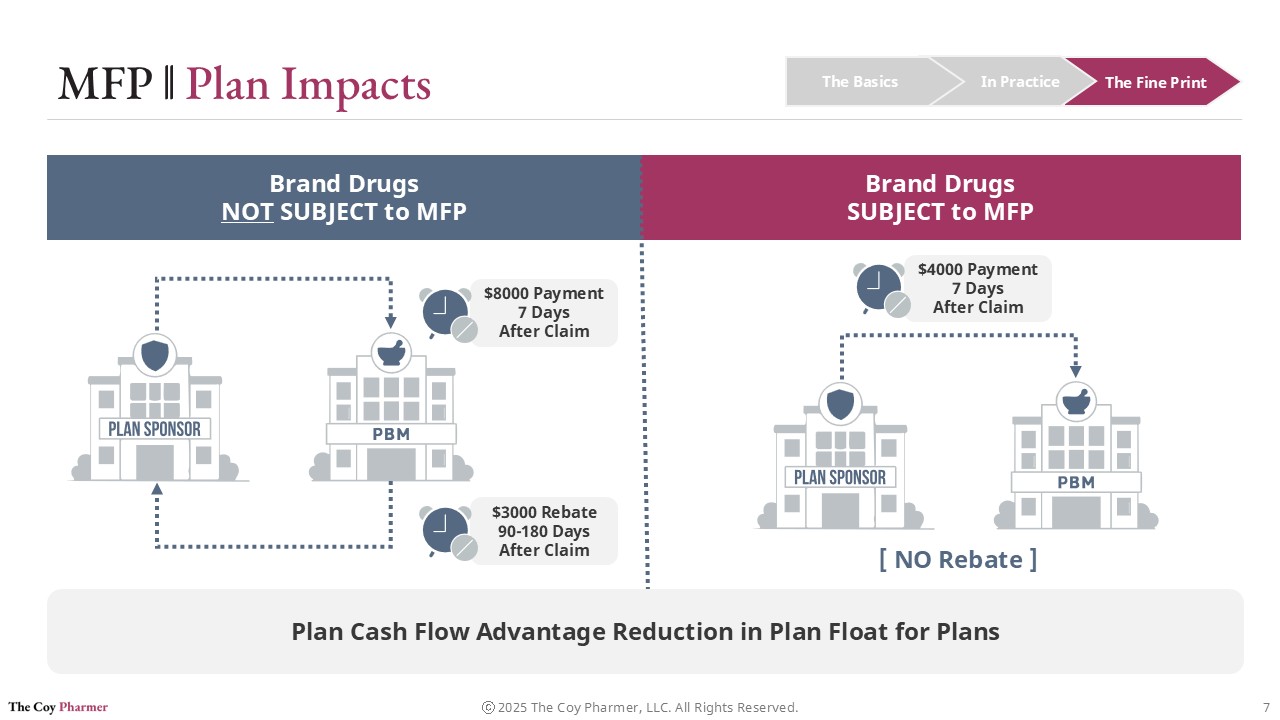
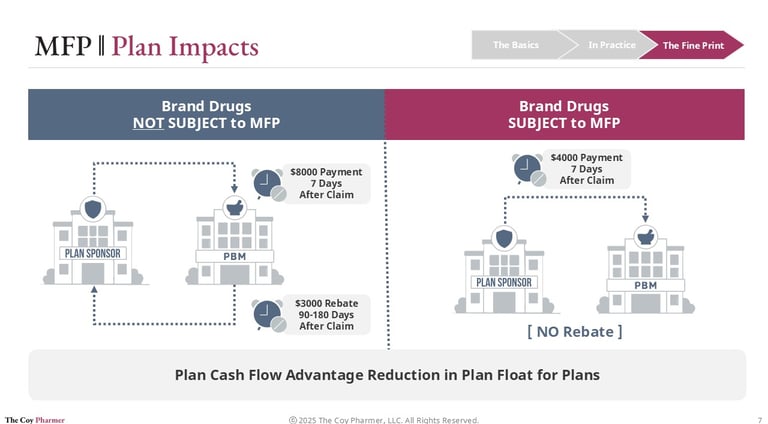
Plan Cash Flow Impacts of MFP
One of the most practical effects of the Maximum Fair Price program is how it changes the timing of dollars moving through the system.
Under the traditional rebate-driven model, plan sponsors typically pay the full negotiated ingredient cost of a brand drug shortly after a claim is processed. That payment may occur days after dispensing, even though a significant portion of the drug’s ultimate net cost will later be offset by manufacturer rebates. Those rebates often arrive months later through reconciliation cycles, creating a gap between when money goes out and when savings come back.
This timing difference can create cash flow pressure for plan sponsors. They must fund the higher up-front cost while waiting for delayed rebate payments to materialize.
For drugs subject to Maximum Fair Price, the dynamic shifts. Instead of paying a high list price up-front and receiving rebates later, payment is based on a lower negotiated price at the point of sale. Pharmacies are reimbursed using this reduced benchmark, which means plan sponsors are billed less from the start.
The result is a movement from a “pay now, reconcile later” model toward a more immediate net pricing structure. While the policy is primarily intended to lower overall spending, the operational effect is improved predictability and reduced float for the entities financing drug claims.
As more high spend products enter the negotiation program, this change in payment timing may become just as consequential as the price reductions themselves.
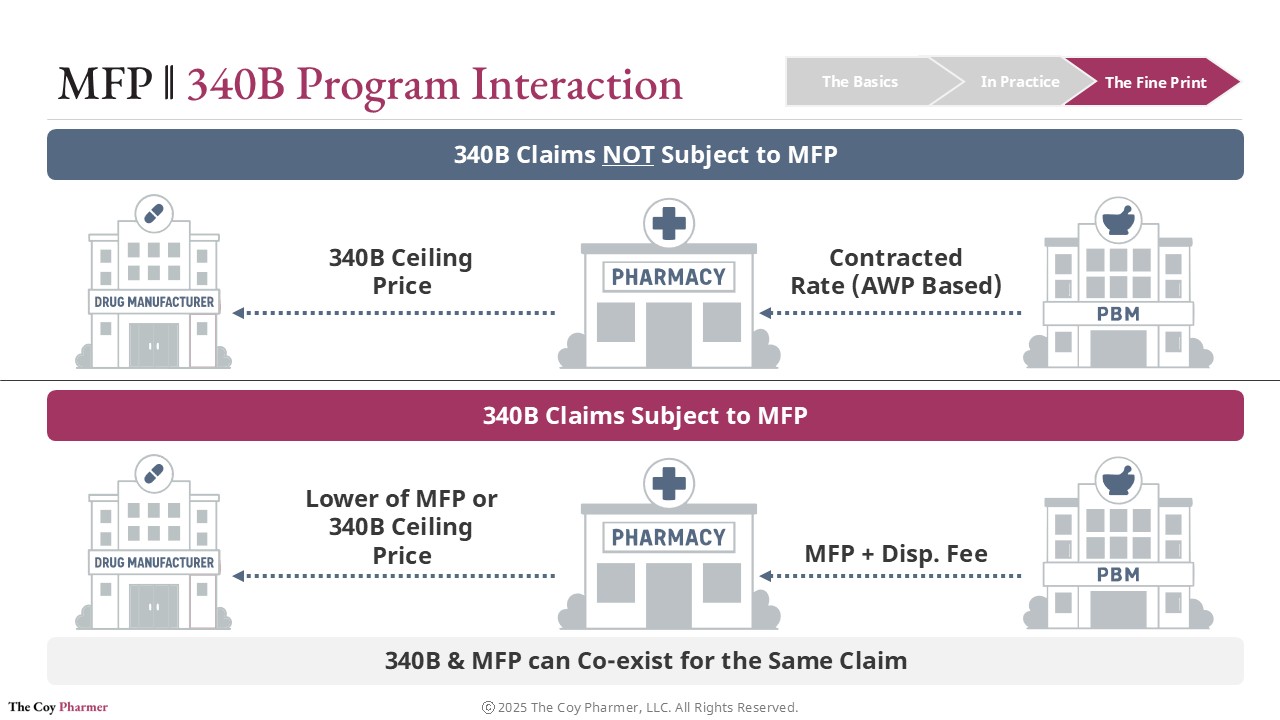
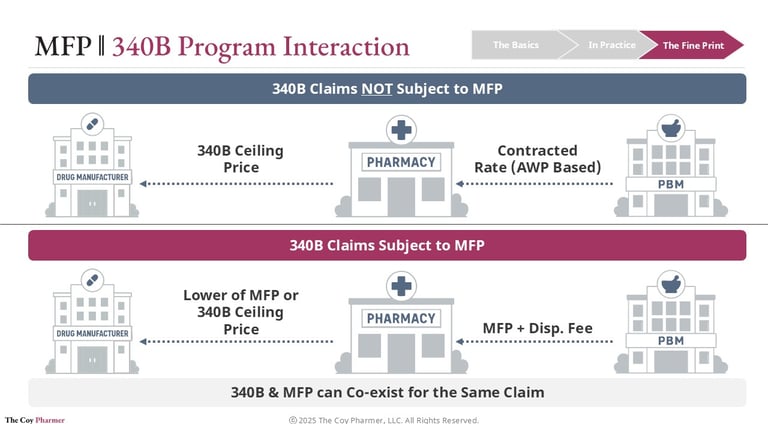
Maximum Fair Price and 340B Program Interaction
When a drug is eligible for both the Medicare negotiation program and the 340B program, the key question becomes how acquisition cost and reimbursement interact.
Under 340B, eligible safety-net providers purchase outpatient drugs at a federally defined 340B Ceiling Price that is typically far below market rates. Health plans or PBMs then reimburse the dispensing pharmacy according to contracted terms that generally do not reflect this lower acquisition cost. The difference between reimbursement and purchase price supports the covered entity’s operations.
If the same drug is also subject to a Maximum Fair Price under Medicare, the acquisition side usually remains unchanged. Covered entities can still obtain the drug at the 340B ceiling price or the MFP if MFP is lower than the 340b ceiling price. The shift occurs on the reimbursement side, where Medicare payment for selected drugs is based on the negotiated Maximum Fair Price.
Because the payment benchmark moves downward, the spread between acquisition cost and reimbursement may shrink. The 340B eligibility itself is not eliminated, but the financial benefit associated with dispensing those drugs can be reduced.
With the shift toward a 340B rebate model, significant complexity could occur. Avalere Health and IQVIA have both published articles an article, outlining the complexity of 340B rebate models interaction with MFP. As more high-spend drugs enter negotiation cycles, this interaction will be an important area for safety-net providers, plans, and policymakers to monitor.
Implications for the Pharmaceutical Industry
MFP introduces direct government pricing power into a market that historically relied on private negotiation and competition. The implications are complex and often debated.
Potential Advantages
Establishes price controls on monopoly-like products
Improves budget predictability for Medicare and taxpayers
May reduce patient out-of-pocket costs for affected drugs
Creates transparency around pricing inputs
Potential Challenges
Applies to a small subset of drugs, limiting overall system savings
Long timelines reduce market flexibility
Manufacturers may adjust launch pricing strategies
Concerns about reduced investment in high-risk R&D
Possible shifts in global pricing behavior
One under-discussed dynamic is behavioral. Manufacturers may accelerate lifecycle strategies such as reformulations, indication expansions, or launch timing decisions to manage exposure to negotiation.
Drug pricing policy rarely stays contained within a single program. Because manufacturers operate globally and across multiple payer types, pricing pressure in Medicare can ripple outward.
Potential downstream effects include:
Cost shifting to commercial markets
Changes in employer plan premiums
Adjustments to rebate strategies
Portfolio reprioritization by manufacturers
Stock market implications for pharmaceutical companies
Large pharmaceutical firms are significant components of major stock indexes and retirement portfolios. Any policy that materially affects revenue streams has broader economic implications beyond healthcare alone.
For independent pharmacies already operating on thin margins, even small changes can have outsized operational consequences.
What Is Maximum Fair Price (MFP)?
Eligibility Criteria for MFP
Links
hello@thecoypharmer.com
© 2025 The Coy Pharmer, LLC. All Rights Reserved.
